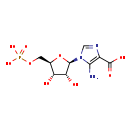| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 23:21:07 UTC |
|---|
| Update Date | 2020-05-21 16:28:53 UTC |
|---|
| BMDB ID | BMDB0006273 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | 5-amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxylate |
|---|
| Description | 5-Amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxylic acid, also known as 1-(5'-phosphoribosyl)-5-amino-4-imidazolecarboxylate, belongs to the class of organic compounds known as pentose phosphates. These are carbohydrate derivatives containing a pentose substituted by one or more phosphate groups. 5-Amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxylic acid exists in all living species, ranging from bacteria to plants to humans. Based on a literature review very few articles have been published on 5-Amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxylic acid. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-(5'-Phosphoribosyl)-4-carboxy-5-aminoimidazole | ChEBI | | 1-(5'-Phosphoribosyl)-5-amino-4-carboxyimidazole | ChEBI | | 1-(5'-Phosphoribosyl)-5-amino-4-imidazolecarboxylate | ChEBI | | 1-(5-Phospho-D-ribosyl)-5-amino-4-imidazolecarboxylate | ChEBI | | 5'-Phosphoribosyl-4-carboxy-5-aminoimidazole | ChEBI | | 5'-Phosphoribosyl-5-amino-4-imidazolecarboxylate | ChEBI | | 5-Amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxylate | ChEBI | | 1-(5'-Phosphoribosyl)-5-amino-4-imidazolecarboxylic acid | Generator | | 1-(5-Phospho-D-ribosyl)-5-amino-4-imidazolecarboxylic acid | Generator | | 5'-Phosphoribosyl-5-amino-4-imidazolecarboxylic acid | Generator | | 5-amino-1-(5-phospho-D-Ribosyl)imidazole-4-carboxylic acid | Generator | | 4-Carboxy-5-aminoimidazole ribonucleotide | HMDB | | 5'Phosphoribosyl-4-carboxy-5-aminoimidazole | HMDB | | 5-Amino-4-carboxyimidazole ribonucleotide | HMDB | | 5-amino-4-Imidazolecarboxylic acid ribonucleotide | HMDB, MeSH | | 5-Aminoimidazole carboxilic acid ribonucleotice | HMDB | | 5-Aminoimidazole-4-carboxilic acid ribonucleotide | HMDB | | Carboxyaminoimidazole ribonucleotide | HMDB | | Carboxy-AIR | MeSH, HMDB | | AICOR | MeSH, HMDB | | Carboxyaminoimidazole ribotide | MeSH, HMDB | | CAIR | MeSH, HMDB | | 5-Amino-1-(5-O-phosphonopentofuranosyl)-1H-imidazole-4-carboxylic acid | MeSH, HMDB | | 1-(5’-Phosphoribosyl)-4-carboxy-5-aminoimidazole | HMDB | | 1-(5’-Phosphoribosyl)-5-amino-4-carboxyimidazole | HMDB | | 1-(5’-Phosphoribosyl)-5-amino-4-imidazolecarboxylate | HMDB | | 5-Amino-1-(5-O-phosphono-beta-D-ribofuranosyl)-1H-imidazole-4-carboxylic acid | HMDB | | 5-Amino-1-(5-O-phosphono-β-D-ribofuranosyl)-1H-imidazole-4-carboxylic acid | HMDB | | 5-Aminoimidazole 4-carboxylic acid ribonucleotide | HMDB | | 5-Aminoimidazolecarboxylic acid ribonucleotide | HMDB | | 5’-Phosphoribosyl-4-carboxy-5-aminoimidazole | HMDB | | 5’-Phosphoribosyl-5-amino-4-imidazolecarboxylate | HMDB |
|
|---|
| Chemical Formula | C9H14N3O9P |
|---|
| Average Molecular Weight | 339.1959 |
|---|
| Monoisotopic Molecular Weight | 339.046765573 |
|---|
| IUPAC Name | 5-amino-1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-[(phosphonooxy)methyl]oxolan-2-yl]-1H-imidazole-4-carboxylic acid |
|---|
| Traditional Name | 5-amino-1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-[(phosphonooxy)methyl]oxolan-2-yl]imidazole-4-carboxylic acid |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | NC1=C(N=CN1[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@H]1O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C9H14N3O9P/c10-7-4(9(15)16)11-2-12(7)8-6(14)5(13)3(21-8)1-20-22(17,18)19/h2-3,5-6,8,13-14H,1,10H2,(H,15,16)(H2,17,18,19)/t3-,5-,6-,8-/m1/s1 |
|---|
| InChI Key | XFVULMDJZXYMSG-ZIYNGMLESA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pentose phosphates. These are carbohydrate derivatives containing a pentose substituted by one or more phosphate groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Pentose phosphates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pentose phosphate
- Pentose-5-phosphate
- Imidazole ribonucleoside
- N-glycosyl compound
- Glycosyl compound
- Monosaccharide phosphate
- Monoalkyl phosphate
- Imidazole-4-carbonyl group
- Alkyl phosphate
- Phosphoric acid ester
- Aminoimidazole
- Organic phosphoric acid derivative
- N-substituted imidazole
- Azole
- Vinylogous amide
- Tetrahydrofuran
- Imidazole
- Heteroaromatic compound
- 1,2-diol
- Secondary alcohol
- Amino acid
- Amino acid or derivatives
- Oxacycle
- Carboxylic acid derivative
- Carboxylic acid
- Azacycle
- Organoheterocyclic compound
- Monocarboxylic acid or derivatives
- Alcohol
- Organic nitrogen compound
- Hydrocarbon derivative
- Amine
- Organopnictogen compound
- Organic oxide
- Primary amine
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-9613000000-2a087bcb710ee7b50b52 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-00lu-2923230000-25e41df497cf1b5c9281 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0914000000-87d6bac417ced888900f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01t9-1900000000-e298883e772e11c253f0 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-8900000000-82583bffa17f1262d918 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004r-8928000000-d296c2812c7b06149794 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9400000000-76793052d8d2699d70dd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-a8b367167e97f6df3690 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0539000000-deee029059e464053ab9 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-2900000000-c7cafadad92d61fbb8a1 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01u0-7900000000-97962f3ed9f96ac07b6a | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-3009000000-ff79299d0fd8156f9b05 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9000000000-a211e114c93b066eaf36 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9100000000-bfdd206a6413421623ed | View in MoNA |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
|
|---|