| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2018-08-29 17:18:13 UTC |
|---|
| Update Date | 2020-06-04 20:27:43 UTC |
|---|
| BMDB ID | BMDB0063201 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | TG(14:1(9Z)/16:1(9Z)/20:2(11Z,14Z))[iso6] |
|---|
| Description | TG(14:1(9Z)/16:1(9Z)/20:2(11Z,14Z))[iso6] belongs to the class of organic compounds known as triacylglycerols. These are glycerides consisting of three fatty acid chains covalently bonded to a glycerol molecule through ester linkages. Thus, TG(14:1(9Z)/16:1(9Z)/20:2(11Z,14Z))[iso6] is considered to be a triradylglycerol lipid molecule. TG(14:1(9Z)/16:1(9Z)/20:2(11Z,14Z))[iso6] is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. |
|---|
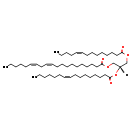
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-(9Z-Tetradecenoyl)-2-(9Z-hexadecenoyl)-3-(11Z,14Z-eicosadienoyl)-glycerol | HMDB | | 1-Myristoleoyl-2-palmitoleoyl-3-eicosadienoyl-glycerol | HMDB | | TAG(14:1/16:1/20:2) | HMDB | | TAG(14:1/16:1/20:2n6) | HMDB | | TAG(14:1/16:1/20:2W6) | HMDB | | TAG(50:4) | HMDB | | TG(14:1/16:1/20:2) | HMDB | | TG(14:1/16:1/20:2n6) | HMDB | | TG(14:1/16:1/20:2W6) | HMDB | | TG(50:4) | HMDB | | Tracylglycerol(14:1/16:1/20:2) | HMDB | | Tracylglycerol(14:1/16:1/20:2n6) | HMDB | | Tracylglycerol(14:1/16:1/20:2W6) | HMDB | | Tracylglycerol(50:4) | HMDB | | Triacylglycerol | HMDB | | Triglyceride | HMDB | | TG(14:1n5/16:1n7/20:2n6) | HMDB | | TG(14:1W5/16:1W7/20:2W6) | HMDB | | Tag(14:1(9Z)/16:1(9Z)/20:2(11Z,14Z)) | HMDB | | Tag(14:1n5/16:1n7/20:2n6) | HMDB | | Tag(14:1W5/16:1W7/20:2W6) | HMDB | | Triacylglycerol(14:1(9Z)/16:1(9Z)/20:2(11Z,14Z)) | HMDB | | Triacylglycerol(14:1/16:1/20:2) | HMDB | | Triacylglycerol(14:1n5/16:1n7/20:2n6) | HMDB | | Triacylglycerol(14:1W5/16:1W7/20:2W6) | HMDB | | Triacylglycerol(50:4) | HMDB | | TG(14:1(9Z)/16:1(9Z)/20:2(11Z,14Z)) | HMDB | | TG(14:1(9Z)/16:1(9Z)/20:2n6) | Lipid Annotator |
|
|---|
| Chemical Formula | C53H94O6 |
|---|
| Average Molecular Weight | 827.329 |
|---|
| Monoisotopic Molecular Weight | 826.705040747 |
|---|
| IUPAC Name | (2R)-2-[(9Z)-hexadec-9-enoyloxy]-3-[(9Z)-tetradec-9-enoyloxy]propyl (11Z,14Z)-icosa-11,14-dienoate |
|---|
| Traditional Name | (2R)-2-[(9Z)-hexadec-9-enoyloxy]-3-[(9Z)-tetradec-9-enoyloxy]propyl (11Z,14Z)-icosa-11,14-dienoate |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | [H][C@@](COC(=O)CCCCCCCCC\C=C/C\C=C/CCCCC)(COC(=O)CCCCCCC\C=C/CCCC)OC(=O)CCCCCCC\C=C/CCCCCC |
|---|
| InChI Identifier | InChI=1S/C53H94O6/c1-4-7-10-13-16-19-22-24-25-26-27-29-31-34-37-40-43-46-52(55)58-49-50(48-57-51(54)45-42-39-36-33-30-21-18-15-12-9-6-3)59-53(56)47-44-41-38-35-32-28-23-20-17-14-11-8-5-2/h15-16,18-20,23-25,50H,4-14,17,21-22,26-49H2,1-3H3/b18-15-,19-16-,23-20-,25-24-/t50-/m0/s1 |
|---|
| InChI Key | BVEHBZABGSQPIA-PFHOFWFYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triacylglycerols. These are glycerides consisting of three fatty acid chains covalently bonded to a glycerol molecule through ester linkages. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Glycerolipids |
|---|
| Sub Class | Triradylcglycerols |
|---|
| Direct Parent | Triacylglycerols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triacyl-sn-glycerol
- Tricarboxylic acid or derivatives
- Fatty acid ester
- Fatty acyl
- Carboxylic acid ester
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Detected and Quantified |
|---|
| Origin | Not Available |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0000000090-2329af1bdf7e46d3ad15 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0000000090-2329af1bdf7e46d3ad15 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0vxr-0000094030-a7c893b68a497cd9d7e8 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0000000090-9bf8c13e2f033a74a3c5 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0000000090-9bf8c13e2f033a74a3c5 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-0000000090-9bf8c13e2f033a74a3c5 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0095080080-40b439d12293bb11ba9f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0032-0093010000-7923946b42dc85936e54 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a6r-3094000000-17bf13027a9fd50e115a | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0000000090-563f171268378ba9fa65 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0000000090-563f171268378ba9fa65 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0vxr-0020094030-288b07237ff17b495f3f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-9530043780-3669db03fceb875daa99 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-9450014700-0f8f933fb3a62edde1cf | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-052u-7381019000-d91d67b30e9bf1aaa06c | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0000000090-ce2f187c72018b25aaf0 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0000000090-ce2f187c72018b25aaf0 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00ai-0040090040-c0ea45176943e48f39e3 | View in MoNA |
|---|
|
|---|